
UNIGADO B
Gadobutrol Injection 604.72 mg/mL (1.0 mmol/mL)
A paramagnetic macrocyclic Gadolinium based contrast agent ( GBCA) for use in MRI
- Gadobutrol (1.0 mmol/mL), an extracellular macrocyclic contrast agent of high stability.
- It is indicated for contrast -enhanced MRI of adults, adolescents and children 2 years of age and older.
- Gadobutrol offers increased T1 relaxivity,which improves MR image quality and clinical use of the technique.
| COMPOSITION | Each ml Gadobutrol Injection contains 604.72 mg/mL (1.0 mmol/mL) of Gadobutrol | |||
|---|---|---|---|---|
| Pack | 7.5ml vial | 15ml vial | 30ml vial | |
| Total Concentration of Gadobutrol | 4.54 g | 9.07 g | 18.14 g | |
| Density (g/ml) at 37°C | 1.30 g/ml | 1.30 g/ml | 1.30 g/ml | |
| Viscosity at 37°C (mPa.s) | 4.96 mPa.s | 4.96 mPa.s | 4.96 mPa.s | |
| Osmolality (mOsmol/kg water) at 37°C | 1603 | 1603 | 1603 | |
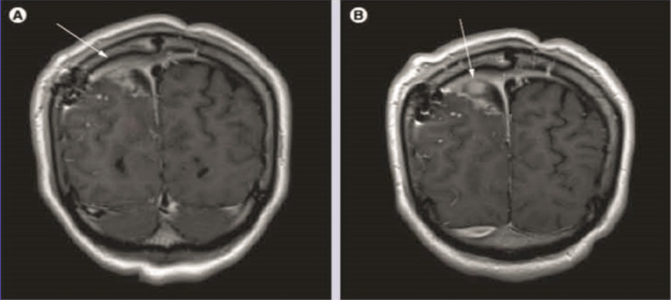
Gadobutrol (A & B) contrast - enhanced spin-echo T-weighted coronal brain images in a patient with brain tumor and postsurgical changes. shows dural enhancement ( A, arrow) at the right and left of the tumor with the presence of nodular area of enhancement ( B, arrow) (A & B) show a better degree of enhancement and internal morphology.
WARNING AND PRECAUTIONS:
Gadolinium-based contrast agents (GBCAs) increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of GBCAs among these patients unless the diagnostic information is essential and not available with non-contrast enhanced MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR <30mL/min/1.73m2) as well as patients with acute kidney injury.
Screen patients for acute kidney injury and other conditions that may reduce renal function.
Before Gadobutrol administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to Gadobutrol. Administer Gadobutrol only in situations where trained personnel and therapies are promptly available for the treatment of hypersensitivity reactions, including personnel trained in resuscitation.
There are no adequate and well-controlled studies of Gadobutrol in pregnant women. While it is unknown if Gadobutrol crosses the human placenta, other gadolinium-based contrast agents (GBCAs) do cross the placenta in humans and result in fetal exposure. Limited published human data on exposure to other GBCAs during pregnancy did not show adverse effects in exposed neonates. Gadobutrol should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Gadobutrol was not teratogenic when given intravenously to monkeys during organogenesis at doses up to 8 times the recommended single human dose (based on body surface area) but was embryolethal at that dose.
ADVERSE REACTION:
The most serious reaction to Gadobutrol are :
i) Nephrogenic systemic fibrosis.
ii) Hypersensitivity reaction.
DOSAGE AND ADMINISTRATION:
Gadobutrol is formulated at a higher concentration (1 mmol/mL) compared to certain other gadolinium based contrast agents, resulting in a lower volume of administration. Closely examine the table below to determine the volume to be administered.
Adults and Children (2 years and older)
The recommended dose of Gadobutrol is 0.1 mL/kg body weight (0.1 mmol/kg).
INDICATIONS:
1. Magnetic Resonance Imaging ( MRI) of the central Nervous System ( CNS):
Gadobutrol is a gadolinium - based contrast agent indicated for intravenous use in diagostic magnetic resonance imaging (MRI) in adults and children (2 years of age and older ) to detect and visualize areas with disrupted blood brain barrier (BBB) and /or abnormal vascularity of the central nervous system.
2. MRI of the Breast:
Gadobutrol is indicated for use with MRI in adults patients to assess the presence and extend of malignant breast disease.
3. Magnetic Resonance Angiography (MRA):
Gadobutrol is indicated for use in magnetic resonance angioghaphy (MRA) in adult and pediatric patients (including term neonates ) to evaluate known or suspected supra-aortic or renal artery disease.
Gadoburtol is indicated for use in cardiac MRI (CMRI) to assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease ( CAD).
CONTRAINDICATION:
Hypersensitivity to Gadobutrol or any product containing gadolinium. Chronic severe kidney disease.
Acute kidney injury.
OVERDOSE:
The maximum dose of Gadobutrol tested in healthy volunteers, 1.5 mL/kg body weight (1.5 mmol/kg) (15 times the recommended dose), was tolerated in a manner similar to lower doses. Gadobutrol can be removed by hemodialysis.
STORAGE:
Store at a temperature not exceeding 30oC. Do not freeze. Protect from light. Keep out of reach of children.
PRESENTATION:
7.5 ml Vial, 15 ml vial, 30 ml vial.
